ELISA Kit
Explore Our Range of ELlSA Kits for veterinary Disease Detection, Vaccine Ethcacy Monitonng,and bisecunty Solutions.
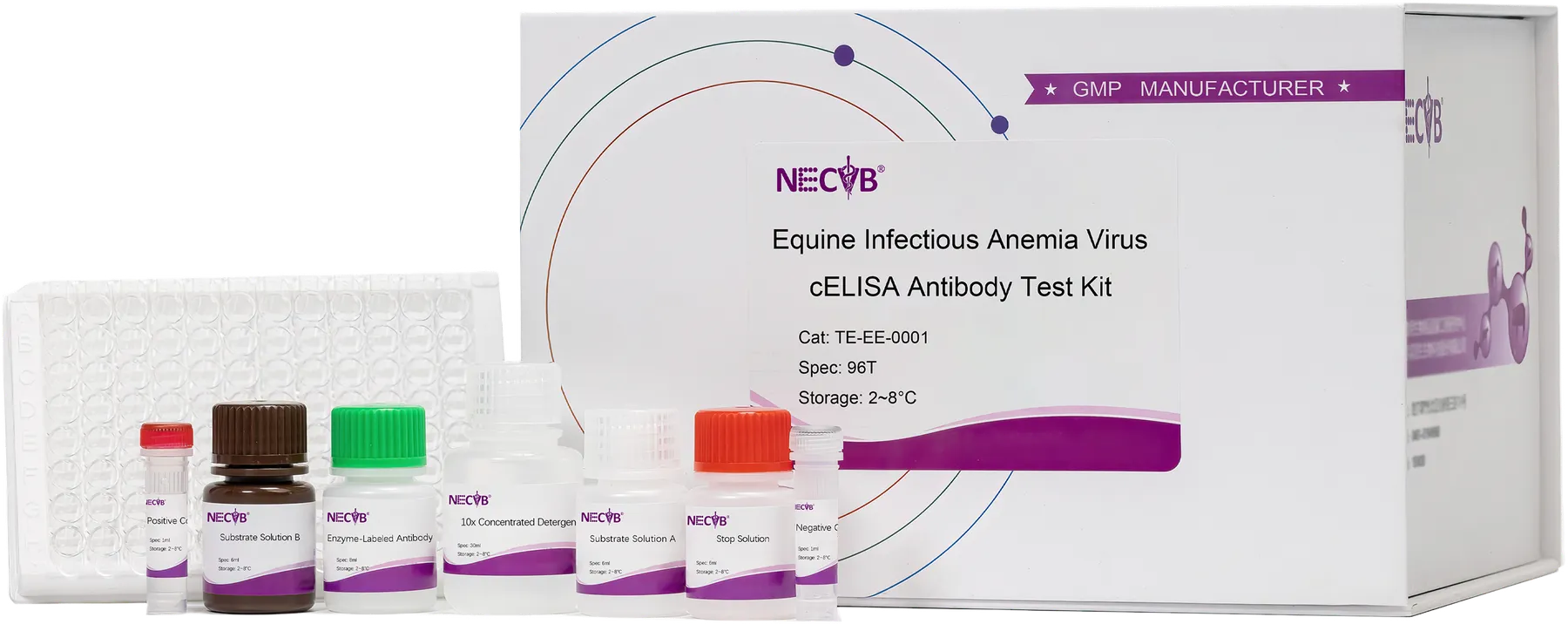
We offer highly sensitive PCR diagnostic kits for animal diseases, specifically designed for accurate pathogen detection. Our products are globally recognized by veterinarians, research institutions, and animal health organizations for their exceptional stability, enabling early diagnosis and comprehensive animal health protection.
| Product Number | TE-FE-0001 |
| Product Name | Equine Infectious Anemia Virus Fluorescent PCR Detection Kit |
| principle | PCR |
| Animal | Equine |
| Specifications | 50T/BOX |
| Test sample | Whole Blood |
| Storage and shelf life | <-20℃,6 months |
| Intended use | The test results of this kit can be used for the auxiliary diagnosis of suspected Equine Infectious Anemia Virus (EIAV) infections |
| G.W./Unit | 0.5kg |
| Certificate | IS09001& GMP-compliant Manufacturing Enterprises & CNAS L12385 |
| 01-Fluorescent PCR reaction mix | 1.15ml/tube*1 |
| 02-Negative control | 100μl/tube*1 |
| 03-Positive control | 30μl/tube*1 |
| 04-Instructions | 1 serving |
Detect target genes with low copy number and minimal cross-reactivity.
Complete detection workflow within 2 hours using standard qPCR machines.
Lyophilized or liquid master mix included, compatible with multiple PCR systems.
Covers bovine, swine, poultry, and companion animals.
For samples with FAM channel Ct value ≤30, report positive for equine infectious anemia virus (EIAV) nucleic acid.
Applicable Instruments: Compatible with ABI series instruments, Roche series instruments, Agilent series instruments, etc.


Detect and control PCV2 spread within farms.

Monitor immune response post-vaccination.

Screen livestock during import/export.

Support academic and field studies.
We will reply you within 24 hours. If for urgent case, please add WhatsApp: +8610522552255 or WeChat:+86 15776899176. Or cal+8610522552255 directly.
*We respect your confidentiality and all information are protected. We will only use your information to respond to your inquiry and will never send unsolicited emails or promotional messages.
We will reply you within 24 hours. If for urgent case, please add WhatsApp/WeChat: +86 19577776076,. Or call +86 15776899176 directly.
*We respect your confidentiality and all information are protected.
We will only use your information to respond to your inquiry and will never send unsolicited emails or promotional messages.